Validation of the Chinese version of the diabetes health profile to predict the impact of mobile health education on quality of life in type 2 diabetes patients
1 Introduction
Type 2 Diabetes Mellitus (T2DM) is a chronic metabolic disorder characterized by insulin resistance, relative insulin deficiency, and hyperglycemia (1). In China, the prevalence of diabetes among adults over 18-years-old has surged to 11.20% (2), triggering severe complications such as heart disease (3–5), stroke (4, 6, 7), kidney disease (3, 8), vision impairment (9, 10) and neuropathy (11). Additionally, it heightens the risk of depression (7), anxiety (12) and other mental health challenges (13). Managing diabetes effectively (14) involves routine blood sugar monitoring (15), adopting a balanced diet (16, 17) and maintaining consistent physical activity (16, 18, 19). These factors profoundly impact patients’ overall quality of life (20, 21), encompassing both their physical and emotional well-being.
1.1 Quality of life in diabetes mellitus
Quality of life (QoL) is a critical health goal that all health interventions strive toward (21). Patients’ perception of their diabetes self-management profoundly influences their QoL, which was often assessed through Patient Self-Reported Outcomes (PROMs) (22, 23). Within PROMs, the evaluating health-related quality of life (HRQOL) holds paramount importance, and various HRQoL questionnaires are utilized in the realm of T2DM (24). HRQOL encompasses a multifaceted construct, encapsulating an individual’s physical, mental and social well-being (25). Unfortunately, China is still in the exploratory phase in this field (26). it is vital to incorporate international diabetes-related scales into the clinical treatment of diabetes in China and adapt them to the local context.
1.2 Diabetes self-care education and assistance
Diabetes Self-management Education and Support (DSMES) play a pivotal role in delivering comprehensive diabetes care (27), aiding patients in understanding and managing their condition, consequently improving health outcomes (28). The Standards for DSMES, which were jointly developed by the American Association of Diabetes Educators and the American Diabetes Association and are updated every 5 years, have proven to enhance the lives of individuals with diabetes through diverse health education initiatives (29). In China, although various diabetes awareness projects exist, yet they often lack coordination and are executed on a limited scale by healthcare professionals (30).
The rapid development of internet and online medical services in the past two decades has fundamentally altered DSMES (31–36). The mobile health (mHealth) model, utilizing mobile and wireless technology encompassing medical and public health services accessible via cellular phones (32), patient monitoring devices (34), personal digital assistants (37), and other wireless devices, aims to optimize health system performance and outcomes through extensive use of Information and Communication Technologies (ICTs). Research has explored the potential of using mobile-based technology to assist individuals with T2DM in maintaining an active lifestyle, enhancing diabetes management, fostering patient-provider communication and offering educational opportunities (27). Implementation of these technologies could empower healthcare providers to tailor patient education, addressing head-on the gaps in the current health education systems.
Notably, a research study conducted in China by Dr. Li Jing and his colleagues underscored the positive physiological and biochemical impacts of the mHealth diabetes management model on individuals with diabetes (34). Based on real-world population data from a clinical electronic health database, they assessed the effectiveness of a mobile-based intervention for glycemic control in patients with T2DM. The research study successfully demonstrated the positive impact of mHealth on blood glucose management. However, its effects on the quality of life among people with diabetes remain uncertain. Further research is essential to confirm whether the mHealth interventions can effectively influence the patient-reported psychosocial outcomes, such as behavioral and activity problems, diabetic misery, dietary inhibition and to comprehend the underlying mechanisms driving these effects.
1.3 The present study
The Diabetes Health Profile 18 (DHP18), which was originally developed for the United Kingdom (36), has been translated into over 30 languages and is now a widely used scale for diabetes-related issues (20, 38–40). In western societies, the DHP18 has been carefully tested and is used in multi-national clinical investigations, HRQOL research, general surveys, and medical practice (41). Although the DHP18 was selected as the diabetes-specific outcome measure for the UK’s PROMs pilot program, there was no modified Chinese version that has been previously developed (42).
Designed for the use in T2DM patients, the DHP18 has demonstrated adequate internal reliability, validity, and measurement equivalence across various language groups (43–45). Its popularity is attributed to several key aspects. Firstly, its brevity with only 18 questions lessens respondent burden, fostering greater patient engagement, particularly suitable for busy Chinese hospital settings. Secondly, the DHP18 delves into multiple mental health domains, encompassing Psychological Distress (PD), Barriers To Activity (BTA), and Disinhibited Eating (DE). Thirdly, none of the existing Chinese instruments cover the DE subscale, despite evidence indicating that DE is a common issue for many people with T2DM (46).
Given all these factors, this study aims to validate a Chinese version of this multidimensional and user-friendly diabetes-related scale and predicts the predicting its impact on Quality of Life (QoL) and the mechanisms of action within Dr. Jing Li’s mHealth model (34).
2 Materials and methods
2.1 Patients
The inclusion criteria for the patients in this study were (i) diagnosed with T2DM, (ii) over 18 years of age and (iii) able to take care of themselves. The data was deleted on account of the following three conditions: short response time (less than 3 min), incomplete questionnaire data and regularity of answers. The study yielded 470 valid responses, with the questionnaire’s validity rate being that of 93.25%. Moreover, 52 T2DM patients who were hospitalized in endocrine wards were selected for retest reliability assessment. This same group of patients underwent repeated measurements at one-month intervals during their hospitalization, serving as the baseline reliability test. All patients were aged between 19 and 79 years, with a mean age of 54 ± 12.40 years.
2.2 Procedure
At first, we developed a paper questionnaire containing all translated DHP18 items, which were pretested to make sure that there were no difficulties in understanding the program. Other relevant questionnaires were also used in this study and included basic information about diabetes (see Table 1 for detailed demographic information). Verbal Informed consent was obtained in three locations: endocrinology wards of the Tianjin People’s Hospital, the Diabetes Identification Center and the outpatient clinics. For ease of sampling, the questionnaires were administered by trained staff to guide patients when they had questions. The Department of Psychology, Nankai University Ethical Review Committee approved the survey.
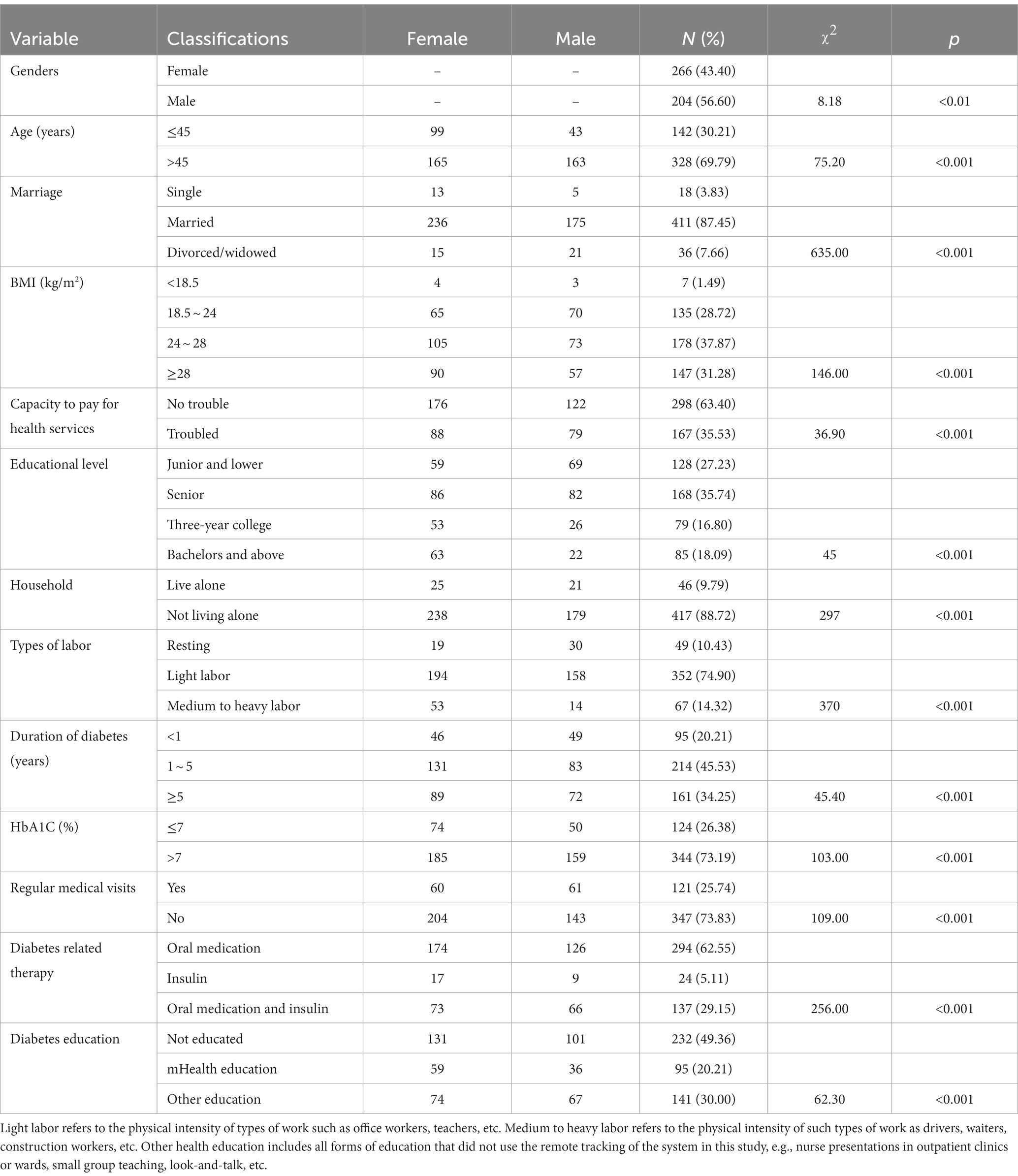
Table 1. Patient demographic variables (N = 470).
2.3 Instruments
2.3.1 Diabetes health profile
The DHP18, initially developed by Meadows et al. (43), comprises of 18 items in three categories: 6 items for PD, 7 items for BTA, and 5 items for DE. Each entry within these categories is evaluated on a Likert 4-point scale, with scores ranging from 0 to 3 for each item. To calculate the total score for each category, the raw scores were added together and divided by the sum of the highest theoretical scores for that category (18, 21, and 15, respectively), then multiplied by 100 to yield a score ranging from 0 to 100. The instrument was translated from English to Chinese by a physician from the Department of Endocrinology at Tianjin People’s Hospital, and then was back-translated from Chinese to English by a professor in the Department of Social Psychology of Tianjin Nankai University. The translated Chinese version was pre-tested to ensure the quality of the scale translation.
2.3.2 EuroQol five-dimensional questionnaire
The EQ-5D-3L, an HRQoL assessment tool developed by Brooks (47), has been translated into a Chinese version and is widely adopted in China (48). It comprises a descriptive system encompassing five health dimensions: mobility, self-care, usual activities, pain or discomfort, and anxiety or depression. Within each dimension, there are three levels denoting the degree of problems: no problems (score 1), some problems (score 2) and extreme problems (score 3). Additionally, we employed the EQ VAS, the vertical visual analog scale evaluated on a 0 to 100-point scale, which was derived using the 2018 Chinese version (49) to measure patients’ quality of life, where higher scores indicate a better quality of life.
2.3.3 The problem areas in diabetes scale
Same as the original version (50), The Chinese version (51) PAID consists of 20 items rated on a Likert 5-point scale, ranging from 0 to 4, where 0 indicates no problem and 4 indicates a severe issue. To derive the total score (ranging from 0 to 100), all item scores are summed and multiplied by 1.25. A total score of 40 or higher suggests emotional distress related to diabetes management, warranting particular attention from healthcare providers.
2.3.4 The appraisal of diabetes scale
ADS was created by Carey et al. (52), and was translated into Chinese by Li et al. (53). It is a seven-item self-evaluation scale that assesses the stress experienced by diabetic patients as a result of their disease. The scale employs a 5-point Likert scoring system, with 5 positively worded items and 2 negatively worded items. The total score is 35, and a lower score indicates that the patient views the disease more positively.
2.3.5 The diabetes self-efficacy scale
The SED, created by Lorig et al. (54) and revised by Wei (55), comprises nine items across four categories in its Chinese version. Scored on a 5-point Likert scale, with 1 indicating a complete lack of confidence and 5 indicating complete confidence, the scale has a total score of 45. Higher scores signify greater confidence in blood glucose management.
2.4 Statistical analysis
All the below mentioned analyses were performed using Jamovi 2.4.8 and Mplus (version 7.4).
2.4.1 Item analysis
Firstly, the top 27% of participants were placed in the high group and the bottom 27% in the low group after computing each participant’s overall score for all items. A t-test was used to compare the differences for each item between the high and low groupings. Secondly, to measure the correlation between each score and the overall score, Pearson’s correlation coefficient was computed. Thirdly, promax rotation was used to further screen items in an exploratory factor analysis (EFA). Prior to conducting the EFA, factor-ality was also evaluated using Bartlett’s test of sphericity and Kaise-Meyer-Olkin (KMO) measures.
2.4.2 Reliability
We calculated Cronbach’s alpha coefficient and retest reliability.
2.4.3 Validity and sensitivity
Confirmatory Factor Analysis (CFA) was performed on each of the three dimensions of the DHP18, and combined reliability and convergent validity were calculated from the factor loadings. We then conducted a test of variability of the DHP18 dimensions across subgroups of other variables to obtain the measurement sensitivity of the scale.
2.4.4 Impacts of mHealth
Given that all the data in this study came from questionnaires, we used Harman’s single-factor test to control common method variance. Then, we calculated the correlation coefficients for every two variables. Additionally, mediation and moderation analyses will be employed to compute and model the correlation between the scores on the various diabetes-related models.
3 Results
3.1 Reliability
The results of the item analysis showed that all the DHP18 items differed at the level of significance on the high and low subgroups, and the correlation coefficients between each item and the total score were also greater than 0.40 and all reached the level of significance (p < 0.001). With a KMO value of 0.87 and a statistically significant Bartlett’s ball test (
= 3,215, df = 153, p < 0.001), the data were suitable for factor analysis. Table 2 illustrates the total Cronbach’s alpha coefficient and test–retest reliability, along with the corresponding values for each dimension of the DHP18. Both the total Cronbach’s alpha coefficient and coefficients of test–retest reliability of the questionnaire exceed 0.80, while each dimension demonstrates Cronbach’s alpha coefficient and coefficients of test–retest reliability surpassing 0.70.
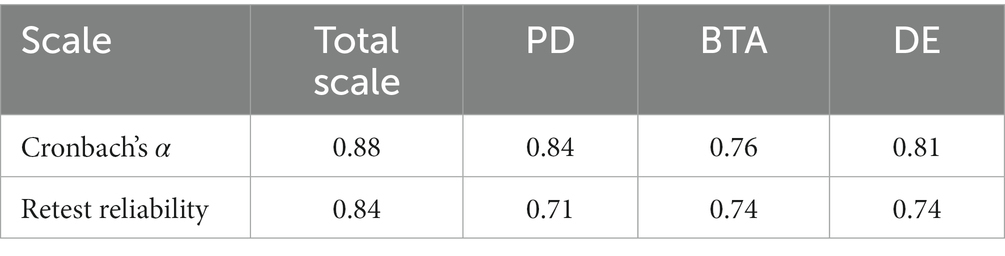
Table 2. Cronbach’s alpha and coefficients of retest reliability.
3.2 Structural validity
CFA was performed on the three dimensions of the DHP18 using Mplus (Version 8.1), and composite reliability and convergent validity were calculated using factor loading. Table 3 shows the results, with CR ranging from 0.77 to 0.85 for each dimension, all of which exceed the 0.60 minimum standard, indicating high reliability of the sample data in each dimension. However, in this study, the Average Variance Extracted (AVE) for the BTA dimension measured 0.33, falling below the factor loading standard of 0.36. Additionally, both the Tucker Lewis Index (TLI) and Comparative Fit Index (CFI) fit indices for the BTA were below 0.90 in the CFA model. These findings suggest that the convergent validity of BTA category of the scale in the present study was not satisfactory and that there is a need to consider further research to understand the necessity of this category and its related indicators.
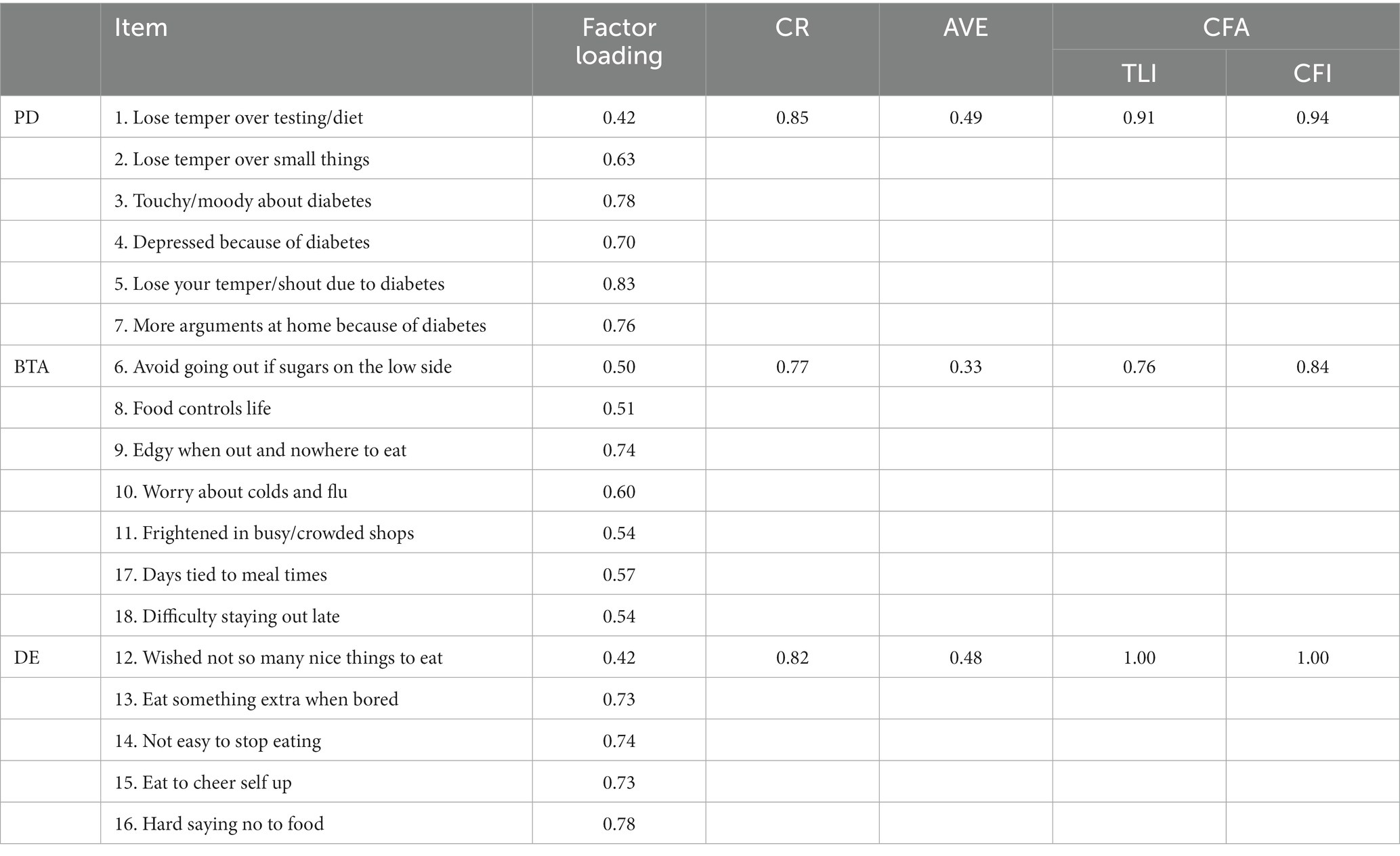
Table 3. Validated factor analysis model fit indices for the sub-dimensional DHP18.
Given the Likert 4-point scoring system utilized by the DHP18 Scale, the MLM estimation method within Mplus was employed to address the possible bias derived from data’s non-normality. Residual correlations between specific items were permitted based on modification indices obtained through the MLM estimation method. The results of the first-order model for the recommended scale, outlined in Tables 4, 5, indicate that all models meet the specified standards.

Table 4. DHP validated factor analysis modified model fit indices.
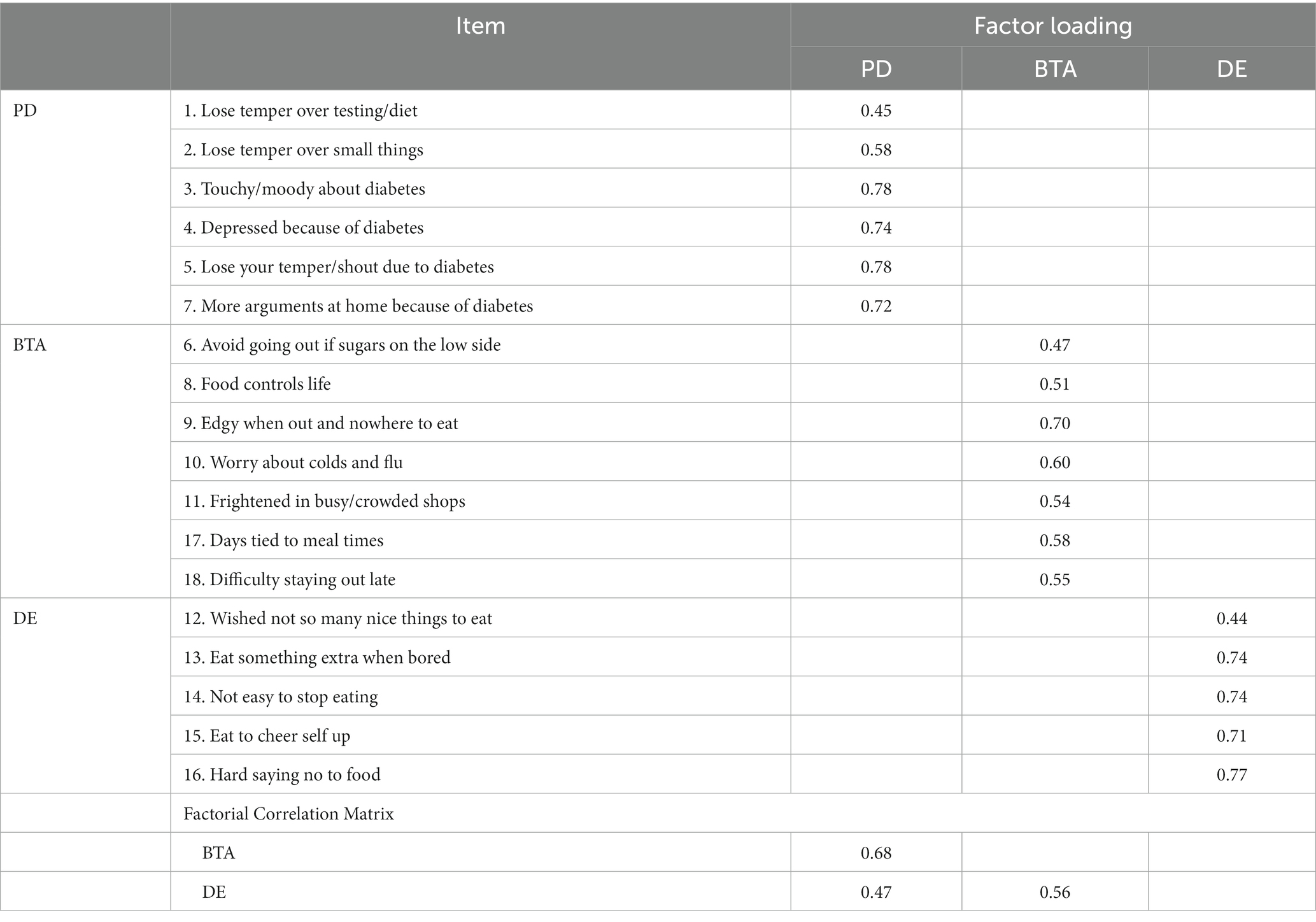
Table 5. DHP18 validated factor analysis modified model factor loading.
3.3 Sensitivity
Table 6 presented the results of sensitivity and known-group validity. The DHP18 demonstrated limited differentiation between genders, educational levels, types of labor, housing situations, and marital status (indicating lower sensitivity) among social determinants. However, it notably showed effectiveness in distinguishing between patients in two different age groups, particularly in measuring PD. Moreover, DE was effective in differentiating between age groups, obese patients, and those within normal BMI ranges. All three subscales—PD, BTA, and DE—proved effective in distinguishing patients based on their ability to afford healthcare, indicating higher sensitivity in this aspect.
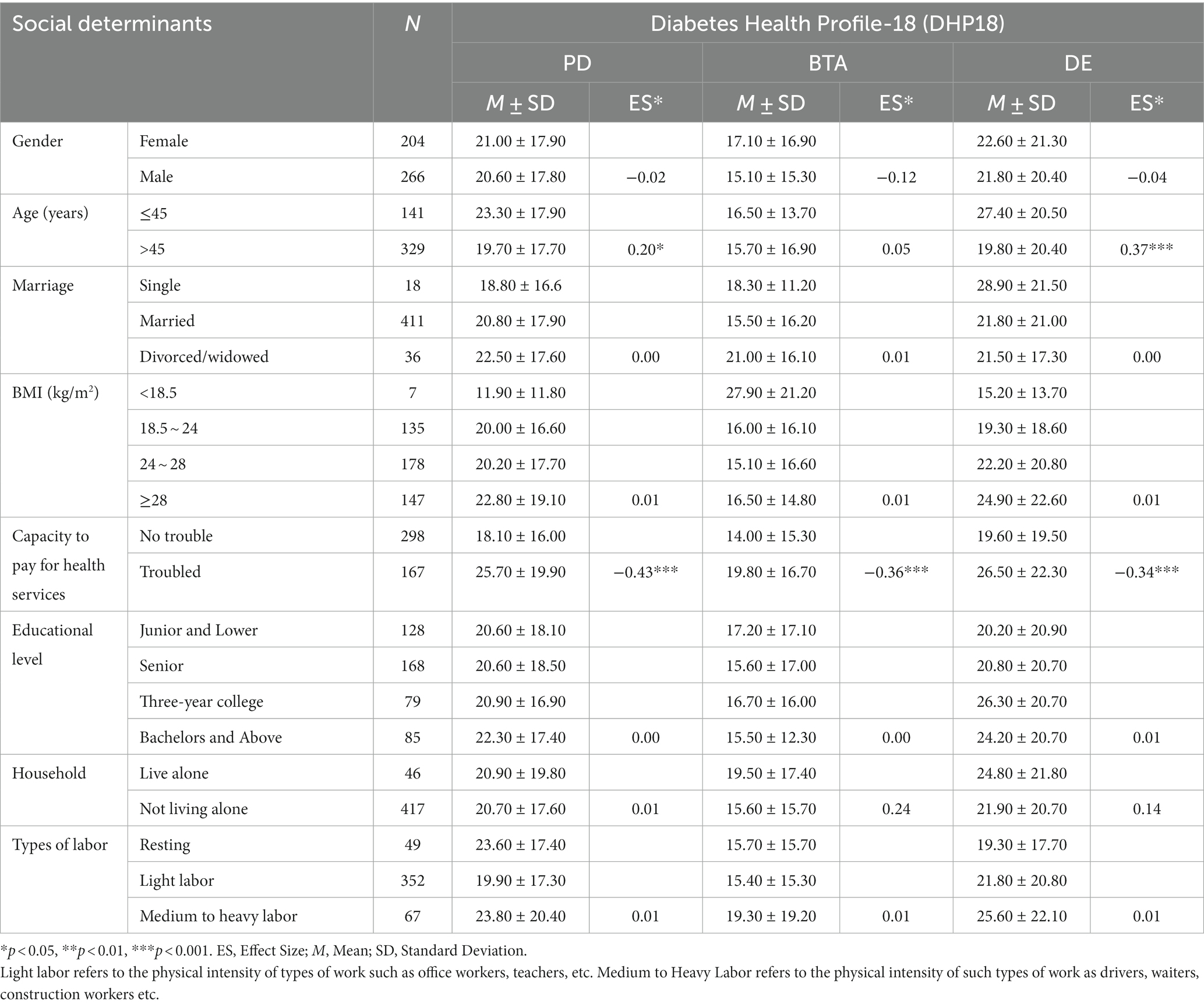
Table 6. Comparison of DHP18 subscales across social determinants.
Table 7 displayed the differential analysis results of DHP18 across various clinical and psychological determinants. Within clinical determinants, the DHP18 subscales exhibited limited capacity to differentiate between patients with varying diabetes durations and different drug treatment methods. Notably, in this survey, only the DE subscale effectively distinguished patients based on their regularity of medical reviews and blood glucose control, using an HbA1c level of 7% as the cutoff point. Regarding psychological determinants, the PAID scores, categorized into two groups based on a total score of 40 points, revealed significant differences in DHP18 subscale scores, indicating a substantial effect size between high and low PAID scores.
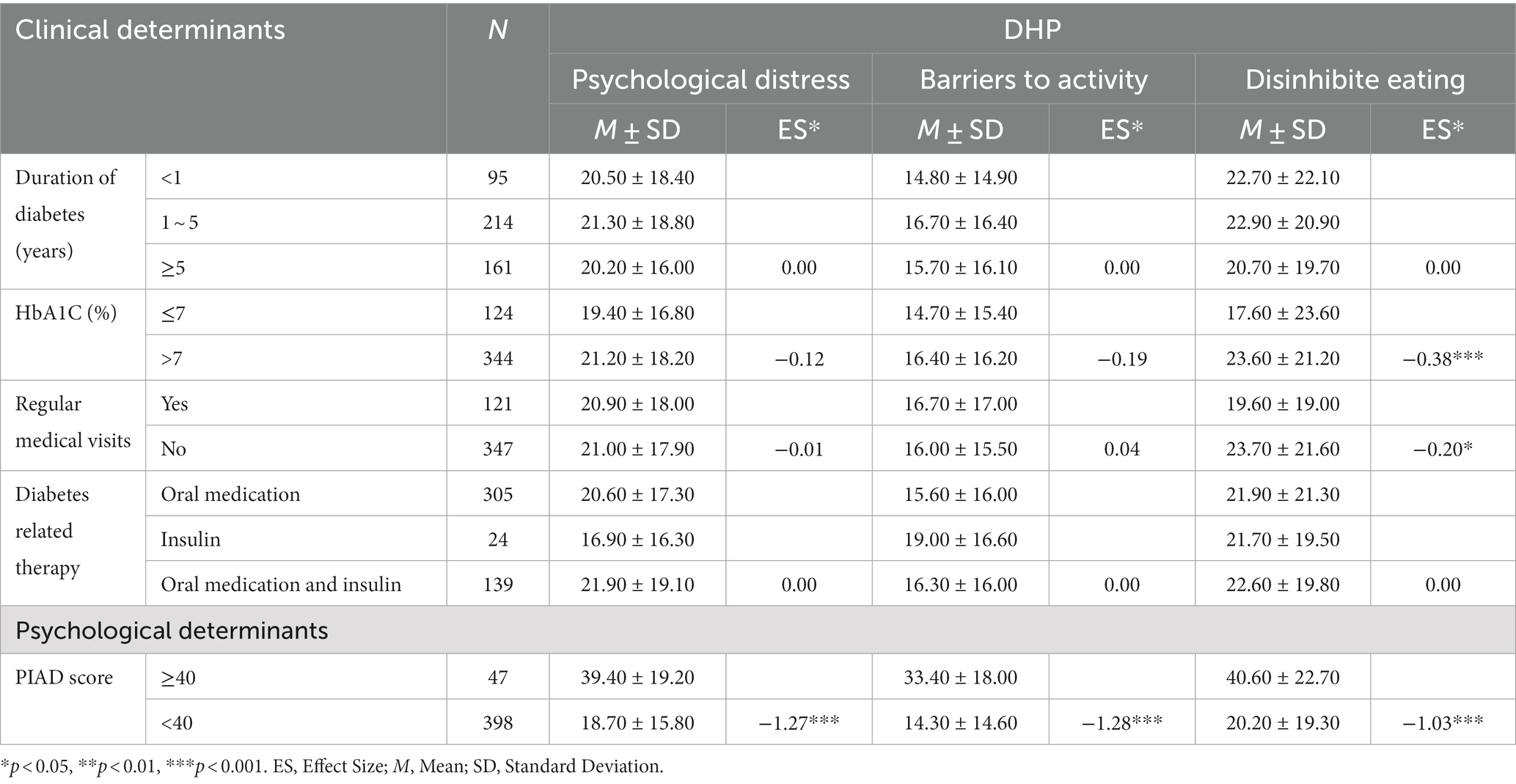
Table 7. Comparison of DHP18 subscales across clinical and psychological determinants.
3.4 Impacts of mHealth
The differences in blood sugar levels (measured by HbA1C) among patients receiving different types of health education were calculated based on the different health education groups. Table 8 showed that all forms of health education, particularly remote care, have a significant positive impact on blood sugar control.
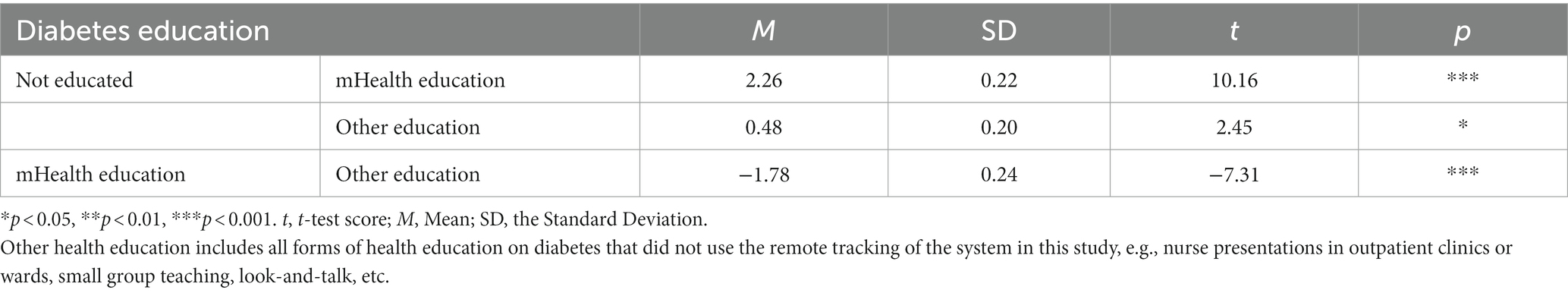
Table 8. Effect of different forms of health education on blood glucose levels.
Table 9 showed that all DHP subscales distinguished well between general diabetes health education (professional talk on diabetes self-management) and mHealth education. The mhealth education included a multidisciplinary team of physicians, nurses, health educators, and dietitians provided continuous, real-time, individualized healthcare through a mobile-based intervention on glycemic control in patients with T2DM. However, no substantial variances in BTA and DE categories were observed between patients who had never received health education and those who were educated online. Furthermore, no statistically significant differences were found between patients who had never received diabetes self-management health education and those who had, with minimal effect sizes observed (PD: p = 0.69, ES = 0.04; BTA: p = 0.07, ES = 0.19; DE: p = 0.26, ES = 0.12).
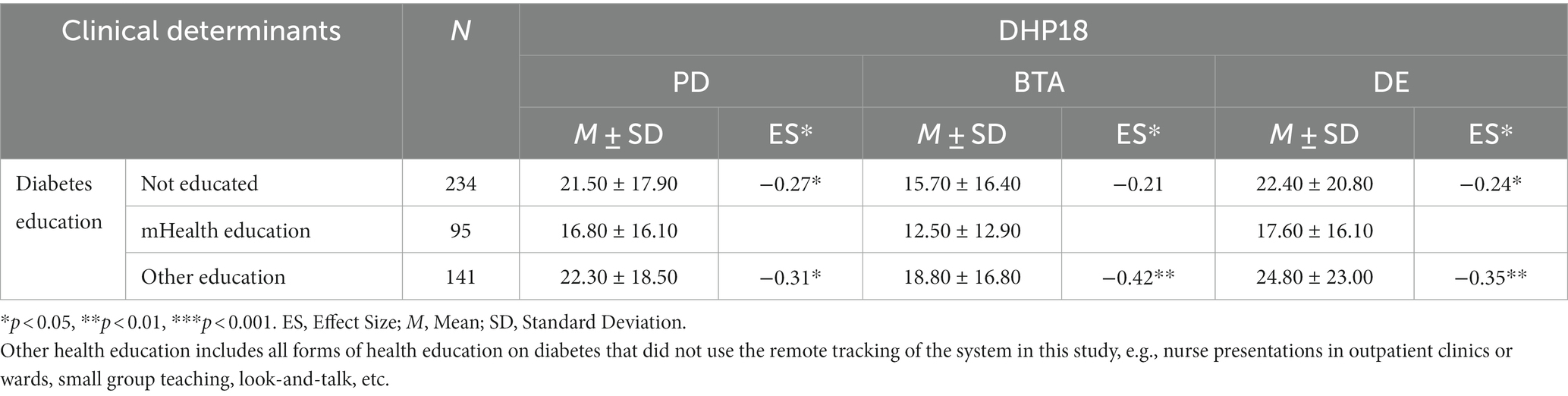
Table 9. Comparison of DHP18 subscales across different forms of health education.
We examined an unrotated EFA solution with a single-factor solution. The first factor loading was 31.89%, which is less than the generally adopted critical standard of 40%. This result indicated the suitability of following mediation and moderation analysis. Table 10 showed the correlations between the different scale dimensions in this study.
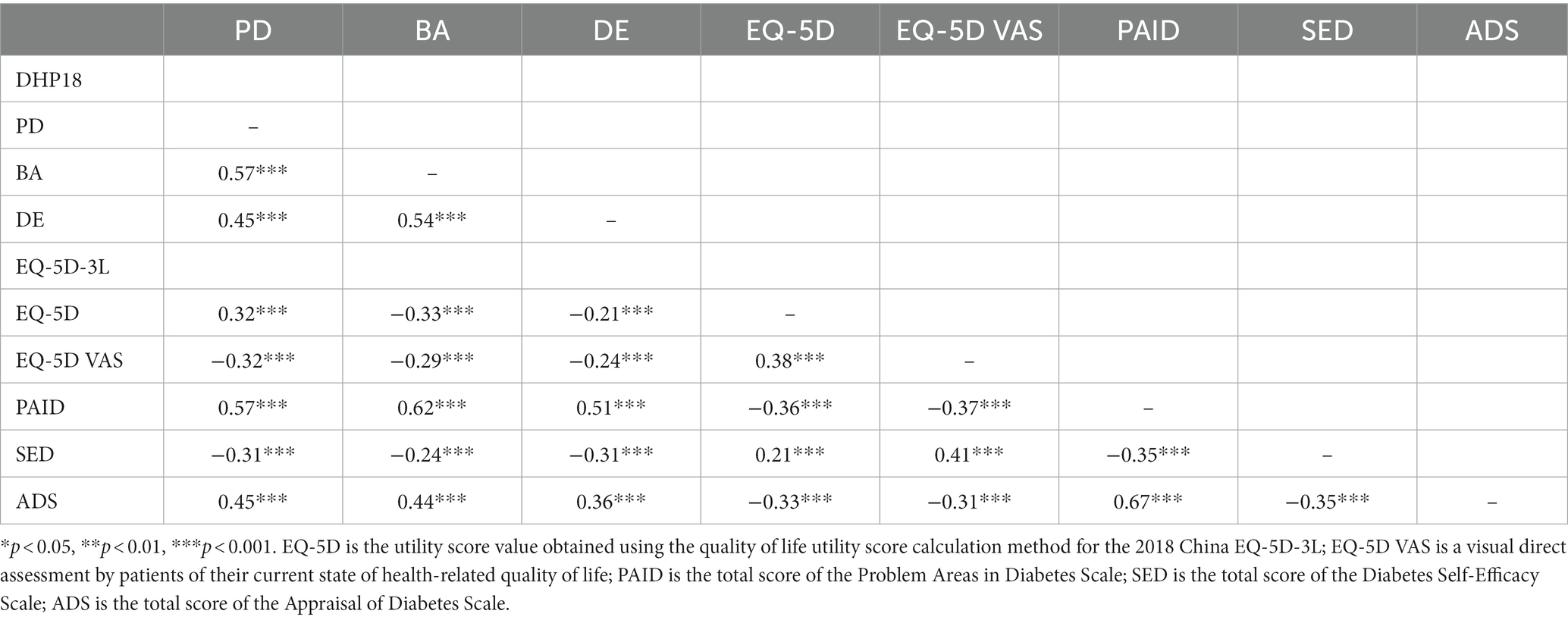
Table 10. Correlations between ADS, SED, EQ-5D-3L and the three dimensions of the DHP18.
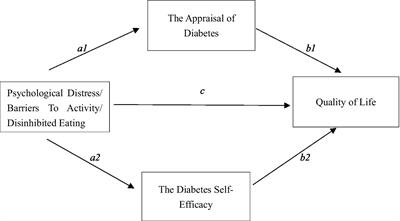
As shown Figure 1, the mediating effects of ADS and SED on the impact of Subscales in the DHP18, including PD, BTA, and DE, on the direct assessment of HRQOL in the EQ-5D-3L VAS were analyzed.
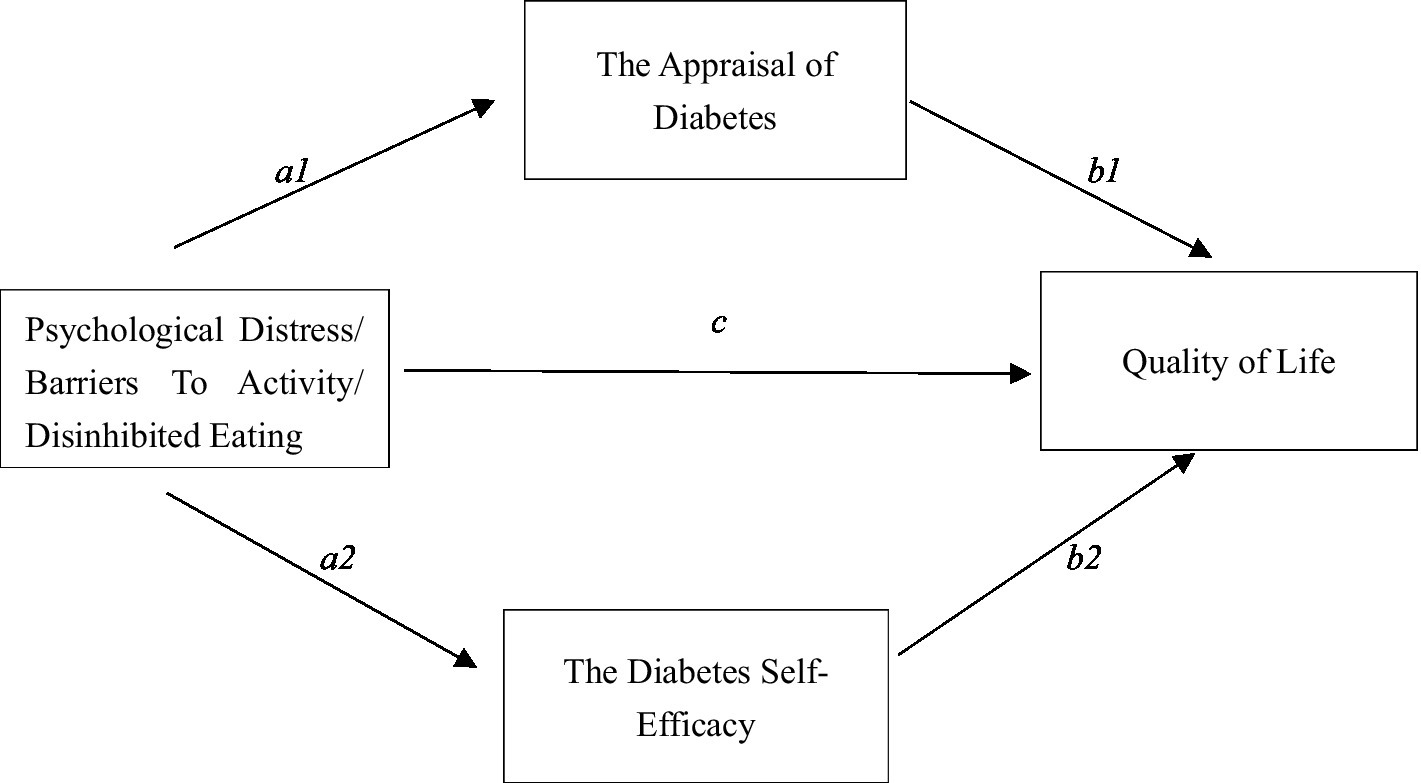
Figure 1. Mediation modeling of DHP18 dimensions on subscales. a1 and b1 are specific indirect effect 1, a2 and b2 are specific indirect effect 2, c is direct effect.
The results are shown in Table 11. The three DHP18 variables significantly underpredicted patient EQ-5D-3L VAS scores for ADS, SED, and QoL. According to the findings of the mediation analysis, ADS and SED were the key mediators in the significant direct and indirect effects of PD and BA on Qol. The BA path was responsible for 49.15% of the total effect, with the PD path’s mediating effect accounting for 47.47% of it.
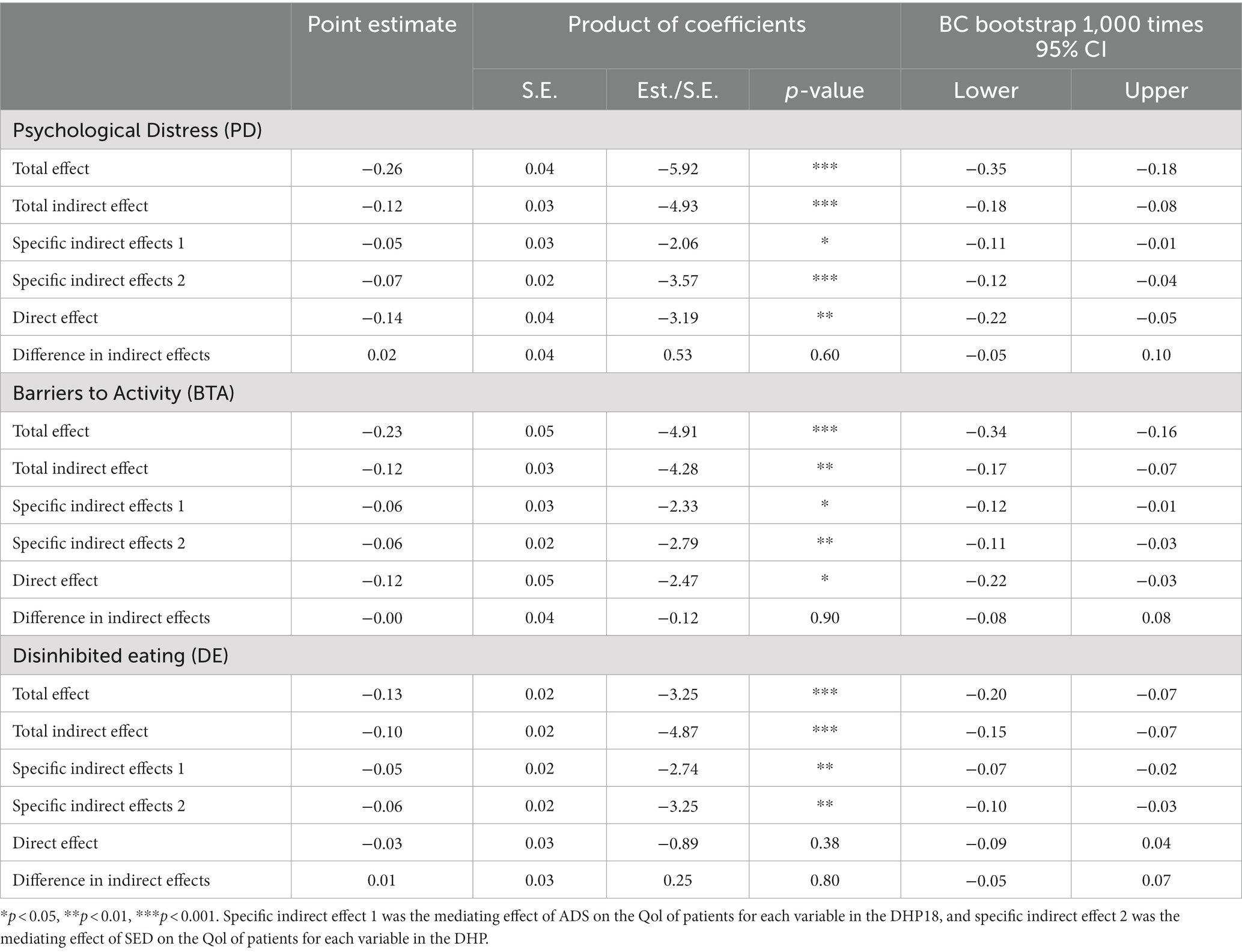
Table 11. Mediating effects of ADS, SED on the three dimensions of the DHP18.
Results of the mediation effect testing model show that the overall effect (β = −0.13, p < 0.001) and the indirect effect (β = −0.10, p < 0.01) of DE on Qol are both significant, but the direct effect is not significant (β = −0.03, p = 0.38). The complete mediating effect of ADS and SED between these two variables is significant, with a total mediation rate of 77.61%, of which ADS accounts for 36.57% and SED accounts for 41.04%. The impact of dietary restraint on Qol is achieved entirely through the indirect pathways of ADS and SED.
4 Discussion
This study validated the effectiveness of the Chinese version of DHP18 and identified some key factors affecting the quality of life of T2DM patients, such as age and economic conditions. Notably, mHealth interventions exhibited a positive impact on the quality of life while also enhancing glycemic control, shedding light on the potential reasons for this favorable outcome.
In this study, the validation of the DHP18 demonstrated its satisfactory validity, reliability, and sensitivity in gaging the psychological well-being of individuals with T2DM. It also had a high criterion validity with another well-studied diabetes-specific distress scale (PAID). We discovered, however, that the BTA subscale has a poor convergent validity, which was consistent with the scale’s validation results in Singapore and Ecuador (44, 45) but differs from its original finding in UK, indicating the possible lacking of the cross-cultural consistency of the structure of DHP18.
Regarding the Effect Sizes (ES), younger diabetic patients (under 45 years) experienced higher psychological distress and greater dietary restriction. Challenges such as availability of healthy meals at work, juggling regular meals and medication, and managing self-health assessments in demanding job roles and the more frequent complications (56) exacerbated stress for younger individuals. Studies indicate that eating behavior might alter under stress (57), potentially explaining the larger effect sizes observed in the DE category (45), which is also applicable to young diabetic patients in China. Moreover, the affordability of diabetes treatment demonstrated significant effect sizes across all three DHP18 dimensions. Limited financial resources correlated with heightened psychological distress (58), impacting behavioral control and dietary management.
Moreover, another important aim of this study is to reveal the underlying mechanism of the mHealth model’s positive function in promoting diabetic patients’ QoL. We highlighted patients’ self-evaluations of diabetes stress and self-efficacy as mediators, and found self-evaluations (59) and self-efficacy (60) can fully mediate the effects of DE on improving patients’ QoL. These results indicate psychological distress, behavioral restrictions and dietary stipulations’ nonnegligible impact on diabetes management. Further analysis on mHealth data showcased the benefits of any form of health education in improving glycemic indicators. Traditional health education often lacked effective measures to monitor patients’ behavioral changes and misconceptions, while the mHealth diabetes management model (34), integrating offline education and remote tracking, empowered patients to apply diabetes knowledge effectively, resolving the misconceptions. The results showed that patients’ own dietary habits have no impact on quality of life; while it is the dietary changes required to manage blood glucose, the inconvenience of living with the behavioral demands of blood glucose self-management, and the emotional distress of diabetes that affect the patients’ quality of life. The usage of mHealth for blood glucose management can provide immediate technical support to alleviate negative emotions from these three areas of DHP18, resulting in positive improvements in self-efficacy and diabetes self-assessment for T2DM patients thus improving their quality of life.
4.1 Implications
This study provides a new Chinese version of the Multidimensional Diabetes-Related Quality of Life Scale for diabetes in China to help healthcare professionals to better tailor mental health education for T2DM patients. Moreover, this study reminds diabetes health educators to focus on special populations and enhance psychological screening for special groups, such as young adults and those with high economic stress. In addition, our findings suggest that online medical education and healthcare play a positive role. The role of mHealth is to curb incorrect glucose control habits more efficiently and accurately. Real-time remote professional guidance from diabetes healthcare professionals can encourage patients to develop a positive concept of diabetes self-management, increase self-efficacy, and improve their evaluation of diabetes, thus improving their quality of life.
4.2 Limitations and future research direction
This study has several limitations. Although the DHP-18 can be used in patients with type 1 or T2DM, the psychometric test was not performed in patients with type 1 diabetes, so the results are only applicable to patients with T2DM. Secondly, due to the cross-sectional nature of this study, there was no cohort follow-up to understand patients’ ongoing quality of life. Thirdly, we investigated patients in only one hospital in Tianjin, China, thus limiting the generalizability of our findings. Multi-center survey and cohort follow-up study are suggested to further prove the psychometric properties and its correlation with other important variables predicting the health condition of patients with T2DM.
5 Conclusion
1. The Chinese version of Diabetes Health Profile Scale has good measurement properties and is appropriate for measuring HRQL in Chinese patients with T2DM.
2. Diabetes health education and support in the mobile health model plays a positive role in the quality of life of people with T2DM.
3. The mHealth model can play a role in improving the quality of life of patients with T2DM by increasing their sense of efficacy in blood glucose self-management and improving their self-assessment of having diabetes.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
XL: Funding acquisition, Writing – review & editing. JZ: Data curation, Formal analysis, Investigation, Writing – original draft. JL: Resources, Supervision, Writing – review & editing. YS: Data curation, Writing – original draft. TY: Data curation, Writing – original draft. WH: Data curation, Writing – original draft.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was partially supported by the National Key Project of Social Sciences of China (20ASH015).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
2. Li, Y
, Teng, D
, Shi, X
, Qin, G
, Qin, Y
, Quan, H, et al. Prevalence of diabetes recorded in mainland China using 2018 diagnostic criteria from the American Diabetes Association: National Cross Sectional Study. BMJ. (2020) 369:m997. doi: 10.1136/bmj.m997
PubMed Abstract | Crossref Full Text | Google Scholar
3. Agarwal, R
, Filippatos, G
, Pitt, B
, Anker, SD
, Rossing, P
, Joseph, A, et al. Cardiovascular and kidney outcomes with finerenone in patients with type 2 diabetes and chronic kidney disease: the Fidelity pooled analysis. Eur Heart J. (2022) 43:1989–9. doi: 10.1093/eurheartj/ehab886
PubMed Abstract | Crossref Full Text | Google Scholar
4. Benjamin, EJ
, Muntner, P
, Alonso, A
, Bittencourt, MS
, Callaway, CW
, Carson, AP, et al. 1. Correction to: heart disease and stroke statistics—2019 update: a report from the American Heart Association. Circulation. (2020) 141:e33. doi: 10.1161/cir.0000000000000746
Crossref Full Text | Google Scholar
6. Ding, G-B
, Sang, Q
, Han, H-J
, Wang, X-M
, and Wu, Y-F
. Assessment of stroke knowledge and awareness among primary healthcare providers: a cross-sectional survey from the Kezhou quality improvement in acute stroke care project. Front Public Health. (2023) 11:170. doi: 10.3389/fpubh.2023.1136170
PubMed Abstract | Crossref Full Text | Google Scholar
7. van Sloten, TT
, Sedaghat, S
, Carnethon, MR
, Launer, LJ
, and Stehouwer, CD
. Cerebral microvascular complications of type 2 diabetes: stroke, cognitive dysfunction, and depression. Lancet Diab Endocrinol. (2020) 8:325–36. doi: 10.1016/s2213-8587(19)30405-x
PubMed Abstract | Crossref Full Text | Google Scholar
8. Bakris, GL
, Agarwal, R
, Anker, SD
, Pitt, B
, Ruilope, LM
, Rossing, P, et al. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med. (2020) 383:2219–29. doi: 10.1056/nejmoa2025845
Crossref Full Text | Google Scholar
9. Steinmetz, JD
, Bourne, RRA
, Briant, PS
, Flaxman, S
, Taylor, HR
, Jonas, JB, et al. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to vision 2020: the right to sight: an analysis for the global burden of disease study. Lancet Global Health. (2021) 9:e144–60. doi: 10.1016/S2214-109x(20)30489-7
PubMed Abstract | Crossref Full Text | Google Scholar
10. Teo, ZL
, Tham, Y-C
, Yu, M
, Chee, ML
, Rim, TH
, Cheung, N, et al. Global prevalence of diabetic retinopathy and projection of burden through 2045 systematic review and Meta-analysis. Ophthalmology. (2021) 128:1580–91. doi: 10.1016/j.ophtha.2021.04.027
PubMed Abstract | Crossref Full Text | Google Scholar
11. Feldman, EL
, Callaghan, BC
, Pop-Busui, R
, Zochodne, DW
, Wright, DE
, Bennett, DL, et al. Diabetic neuropathy. Nat Rev Dis Prim. (2019) 5:41. doi: 10.1038/s41572-019-0092-1
Crossref Full Text | Google Scholar
13. Robinson, DJ
, Coons, M
, Haensel, H
, Vallis, M
, and Yale, J-F
. Diabetes and mental health. Can J Diabetes. (2018) 42:S130–41. doi: 10.1016/j.jcjd.2017.10.031
Crossref Full Text | Google Scholar
14. Zhang, Y
, Pan, X-F
, Chen, J
, Xia, L
, Cao, A
, Zhang, Y, et al. Combined lifestyle factors and risk of incident type 2 diabetes and prognosis among individuals with type 2 diabetes: a systematic review and meta-analysis of prospective cohort studies. Diabetologia. (2019) 63:21–33. doi: 10.1007/s00125-019-04985-9
PubMed Abstract | Crossref Full Text | Google Scholar
15. Martina, M
, Marco, C
, Davide, G
, and Giuseppe, L
. Overview on self-monitoring of blood glucose. Clin Chim Acta. (2009) 402:7–13. doi: 10.1016/j.cca.2009.01.002
Crossref Full Text | Google Scholar
16. Magkos, F
, Hjorth, MF
, and Astrup, A
. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. (2020) 16:545–55. doi: 10.1038/s41574-020-0381-5
Crossref Full Text | Google Scholar
17. Xu, Z
, Steffen, LM
, Selvin, E
, and Rebholz, CM
. Diet quality, change in diet quality and risk of incident CVD and diabetes. Public Health Nutr. (2020) 23:329–38. doi: 10.1017/s136898001900212x
PubMed Abstract | Crossref Full Text | Google Scholar
18. Amanat, S
, Ghahri, S
, Dianatinasab, A
, Fararouei, M
, and Dianatinasab, M
. Exercise and type 2 diabetes. Phys Exerc Hum Health. (2020) 1228:91–105. doi: 10.1007/978-981-15-1792-1_6
Crossref Full Text | Google Scholar
19. Kanaley, JA
, Colberg, SR
, Corcoran, MH
, Malin, SK
, Rodriguez, NR
, Crespo, CJ, et al. Exercise/physical activity in individuals with type 2 diabetes: a consensus statement from the American College of Sports Medicine. Med Sci Sports Exerc. (2022) 54:353–68. doi: 10.1249/mss.0000000000002800
PubMed Abstract | Crossref Full Text | Google Scholar
20. Co, MA
, Tan, LSM
, Tai, ES
, Griva, K
, Amir, M
, Chong, KJ, et al. Factors associated with psychological distress, behavioral impact and health-related quality of life among patients with type 2 diabetes mellitus. J Diabetes Complicat. (2015) 29:378–83. doi: 10.1016/j.jdiacomp.2015.01.009
PubMed Abstract | Crossref Full Text | Google Scholar
21. Rubin, RR
, and Peyrot, M
. Quality of life and diabetes. Diabetes Metab Res Rev. (1999) 15:205–18. doi: 10.1002/(sici)1520-7560(199905/06)15:3<205::aid-dmrr29>3.0.co;2-o
Crossref Full Text | Google Scholar
22. Rubin, RR
, Peyrot, M
, and Siminerio, LM
. Health care and patient-reported outcomes: results of the cross-national diabetes attitudes, wishes and needs (DAWN) study. Diabetes Care. (2006) 29:1249–55. doi: 10.2337/dc05-2494
Crossref Full Text | Google Scholar
23. Wee, PJL
, Kwan, YH
, Loh, DHF
, Phang, JK
, Puar, TH
, Østbye, T, et al. Measurement properties of patient-reported outcome measures for diabetes: systematic review. J Med Internet Res. (2021) 23:e25002. doi: 10.2196/25002
PubMed Abstract | Crossref Full Text | Google Scholar
24. Langendoen-Gort, M
, Groeneveld, L
, Prinsen, CAC
, Beulens, JW
, Elders, PJM
, Halperin, I, et al. Patient-reported outcome measures for assessing health-related quality of life in people with type 2 diabetes: a systematic review. Rev Endocrine Metab Disord. (2022) 23:931–77. doi: 10.1007/s11154-022-09734-9
PubMed Abstract | Crossref Full Text | Google Scholar
25. Squitieri, L
, Bozic, KJ
, and Pusic, AL
. The role of patient-reported outcome measures in value-based payment reform. Value Health. (2017) 20:834–6. doi: 10.1016/j.jval.2017.02.003
PubMed Abstract | Crossref Full Text | Google Scholar
26. Wu, J-Y
, Ning, L
, and Tang, A-M
. Advances in clinical applications of reported outcomes in patients with diabetes mellitus. Nurs J Chinese People’s Liberation Army. (2020) 37:857–60. doi: 10.3969/j.issn.1008-9993.2020.08.014
Crossref Full Text | Google Scholar
27. ElSayed, NA
, Aleppo, G
, Aroda, VR
, Bannuru, RR
, Brown, FM
, Bruemmer, D, et al. 5. Facilitating positive health behaviors and well-being to improve health outcomes: standards of care in diabetes—2023. Diabetes Care. (2023) 46:S68–96. doi: 10.2337/dc23-S005
PubMed Abstract | Crossref Full Text | Google Scholar
28. Powers, MA
, Bardsley, J
, Cypress, M
, Duker, P
, Funnell, MM
, Fischl, AH, et al. Diabetes self-management education and support in type 2 diabetes. Diabetes Educ. (2017) 41:417–30. doi: 10.1177/0145721715588904
Crossref Full Text | Google Scholar
29. Davis, J
, Fischl, AH
, Beck, J
, Browning, L
, Carter, A
, Condon, JE, et al. 2022 national standards for diabetes self-management education and support. Sci Diab Manag Care. (2022) 48:44–59. doi: 10.1177/26350106211072203
PubMed Abstract | Crossref Full Text | Google Scholar
30. Deng, H-J
, and Li, W-D
. Review of community health education on patients with type 2 diabetes. Chinese J Health Educ. (2017) 33:7. doi: 10.16168/j.cnki.issn.1002-9982.2017.07.016
Crossref Full Text | Google Scholar
31. Cornford, T
, Free, C
, Phillips, G
, Galli, L
, Watson, L
, Felix, L, et al. The effectiveness of mobile-health technology-based health behaviour change or disease management interventions for health care consumers: a systematic review. PLoS Med. (2013) 10:e1001362. doi: 10.1371/journal.pmed.1001362
PubMed Abstract | Crossref Full Text | Google Scholar
32. Agarwal, S
, LeFevre, AE
, Lee, J
, L’Engle, K
, Mehl, G
, Sinha, C, et al. Guidelines for reporting of health interventions using mobile phones: mobile health (mHealth) evidence reporting and assessment (mERA) checklist. BMJ. (2016) 352:i1174. doi: 10.1136/bmj.i1174
PubMed Abstract | Crossref Full Text | Google Scholar
33. Kayyali, R
, Peletidi, A
, Ismail, M
, Hashim, Z
, Bandeira, P
, and Bonnah, J
. Awareness and use of mHealth apps: a study from England. Pharmacy. (2017) 5:33. doi: 10.3390/pharmacy5020033
PubMed Abstract | Crossref Full Text | Google Scholar
34. Li, J
, Sun, L
, Wang, Y
, Guo, L
, Li, D
, Liu, C, et al. A mobile-based intervention for glycemic control in patients with type 2 diabetes: retrospective, propensity score-matched cohort study. JMIR Mhealth Uhealth. (2020) 8:e15390. doi: 10.2196/15390
PubMed Abstract | Crossref Full Text | Google Scholar
35. McCool, J
, Dobson, R
, Whittaker, R
, and Paton, C
. Mobile health (mHealth) in low- and middle- income countries. Annu Rev Public Health. (2022) 43:525–39. doi: 10.1146/annurev-publhealth-052620-093850
Crossref Full Text | Google Scholar
36. Jansky, B
, Machleid, F
, and Wild, V
. Mobile health technologies, social justice and population-based vulnerabilities a public health ethics perspective on mHealth using the example of type 2 diabetes mellitus. Bundesgesundheitsblatt-Gesundheitsforschung-Gesundheitsschutz. (2023) 66:168–75. doi: 10.1007/s00103-022-03650-8
Crossref Full Text | Google Scholar
37. Sevick, MA
, Zickmund, S
, Korytkowski, M
, Piraino, B
, Sereika, S
, Mihalko, S, et al. Design, feasibility, and acceptability of an intervention using personal digital assistant-based self-monitoring in managing type 2 diabetes. Contemp Clin Trials. (2008) 29:396–409. doi: 10.1016/j.cct.2007.09.004
PubMed Abstract | Crossref Full Text | Google Scholar
38. Garratt, AM
, Schmidt, L
, and Fitzpatrick, R
. Patient-assessed health outcome measures for diabetes: a structured review. Diabet Med. (2002) 19:1–11. doi: 10.1046/j.1464-5491.2002.00650.x
PubMed Abstract | Crossref Full Text | Google Scholar
39. Baron, JS
, Hirani, S
, and Newman, SP
. A randomised, controlled trial of the effects of a mobile telehealth intervention on clinical and patient-reported outcomes in people with poorly controlled diabetes. J Telemed Telecare. (2016) 23:207–16. doi: 10.1177/1357633×16631628
PubMed Abstract | Crossref Full Text | Google Scholar
40. Yeoh, E
, Tan, SG
, Lee, YS
, Tan, HH
, Low, YY
, Lim, SC, et al. Impact of COVID-19 and partial lockdown on access to care, self-management and psychological well-being among people with diabetes: a cross-sectional study. Int J Clin Pract. (2021) 75:e14319. doi: 10.1111/ijcp.14319
PubMed Abstract | Crossref Full Text | Google Scholar
41. Mulhern, B
, and Meadows, K. Estimating the minimally important difference (MID) of the diabetes health Profile-18 (DHP-18) for type 1 and type 2 diabetes mellitus. Discussion Paper (2011).
Google Scholar
42. Zhang, GGYD
. Progress in the diabetes-specific quality of survival scale. Chin J Prev Contr Chron Non-commun Dis. (2005) 13:313–5. doi: 10.3969/j.issn.1004-6194.2005.06.028
Crossref Full Text | Google Scholar
43. Meadows, KA
, Abrams, C
, and Sandbaek, A
. Adaptation of the diabetes health profile (DHP-1) for use with patients with type 2 diabetes mellitus: psychometric evaluation and cross-cultural comparison. Diabet Med. (2000) 17:572–80. doi: 10.1046/j.1464-5491.2000.00322.x
PubMed Abstract | Crossref Full Text | Google Scholar
44. Benazizi, I
, Bernal-Soriano, MC
, Pardo, Y
, Ribera, A
, Peralta-Chiriboga, A
, Ferrer, M, et al. Adaptation and psychometric validation of diabetes health profile (DHP-18) in patients with type 2 diabetes in Quito, Ecuador: a cross-sectional study. Health Qual Life Outcomes. (2021) 19:189. doi: 10.1186/s12955-021-01818-5
PubMed Abstract | Crossref Full Text | Google Scholar
45. Tan, MLKE
, Griva, K
, Lee, YS
, Amir, M
, Zuniga, YL
, Lee, J, et al. Diabetes health Profile-18 is reliable, valid and sensitive in Singapore. Ann Acad Med Singap. (2016) 45:383–93. doi: 10.47102/annals-acadmedsg.V45N9p383
PubMed Abstract | Crossref Full Text | Google Scholar
46. Ouwens, MA
, van Strien, T
, and van der Staak, CPF
. Tendency toward overeating and restraint as predictors of food consumption. Appetite. (2003) 40:291–8. doi: 10.1016/s0195-6663(03)00006-0
PubMed Abstract | Crossref Full Text | Google Scholar
48. Wang, H-M
, Patrick, DL
, Edwards, TC
, Skalicky, AM
, Zeng, H-Y
, and Gu, W-W
. Validation of the EQ-5D in a general population sample in urban China. Qual Life Res. (2011) 21:155–60. doi: 10.1007/s11136-011-9915-6
PubMed Abstract | Crossref Full Text | Google Scholar
49. Zhuo, L
, Xu, L
, Ye, J
, Sun, S
, Zhang, Y
, Burstrom, K, et al. Time trade-off value set for EQ-5D-3L based on a nationally representative chinese population survey. Value Health. (2018) 21:1330–7. doi: 10.1016/j.jval.2018.04.1370
PubMed Abstract | Crossref Full Text | Google Scholar
50. William, H
, Polonsky, BJA
, Lohrer, PA
, Welch, G
, Jacobson, AM
, Aponte, JE, et al. Assessment of diabetes-related distress. Diabetes Care. (1995) 18:754–60. doi: 10.2337/diacare.18.6.754
Crossref Full Text | Google Scholar
51. Huang, M-F
, Courtney, M
, Edwards, H
, and McDowell, J
. Validation of the chinese version of the problem areas in diabetes (PAID-C) scale. Diabetes Care. (2010) 33:38–40. doi: 10.2337/dc09-0768
PubMed Abstract | Crossref Full Text | Google Scholar
52. Carey, MP
, Jorgensen, RS
, Weinstock, RS
, Jorgensen, RS
, Weinstock, RS
, Sprafkin, RP, et al. Reliability and validity of the appraisal of diabetes scale. J Behav Med. (1991) 14:43–50. doi: 10.1007/BF00844767
Crossref Full Text | Google Scholar
53. Cheng, L
, Li, X-M
, Gao, R
, Hui, RS
, Yang, J-L
, Wu, Y-N, et al. The reliability and validity of the Chinese version of the appraisal of diabetes scale. Chin J Nurs Educ. (2013) 10:485–8. doi: 10.3761/j.issn.1672-9234.2013.11.002
Crossref Full Text | Google Scholar
55. Hu, J-J
, Dong, Y
, Wei, J
, and Huang, X-H
. Correlation between self-efficacy, self-management and glycemic control among seniors with diabetes in rural areas. Chin Prev Med. (2013) 11:832–6. doi: 10.16506/j.1009-6639.2013.11.013
Crossref Full Text | Google Scholar
56. Middleton, TL
, Constantino, MI
, Molyneaux, L
, D’Souza, M
, Twigg, SM
, Wu, T, et al. Young-onset type 2 diabetes and younger current age: increased susceptibility to retinopathy in contrast to other complications. Diabet Med. (2020) 37:991–9. doi: 10.1111/dme.14238
PubMed Abstract | Crossref Full Text | Google Scholar
57. Weidner, G
, Kohlmann, CW
, Dotzauer, E
, and Burns, LR
. The effects of academic stress on health behaviors in young adults. Anxiety Stress Coping. (1996) 9:123–33. doi: 10.1080/10615809608249396
Crossref Full Text | Google Scholar
58. Pahlevan Sharif, S
, Naghavi, N
, Ong, FS
, Sharif Nia, H
, and Waheed, H
. Health insurance satisfaction, financial burden, locus of control and quality of life of cancer patients: a moderated mediation model. Int J Soc Econ. (2021) 48:513–30. doi: 10.1108/IJSE-10-2019-0629
Crossref Full Text | Google Scholar
59. Wong, JJ
, Hanes, SJ
, Flores, H
, Ngo, J
, and Hood, KK
. Satisfaction with diabetes involvement: exploring parent and adolescent perspectives. Diabet Med. (2023):e15254. doi: 10.1111/dme.15254
PubMed Abstract | Crossref Full Text | Google Scholar
60. Juarez, LD
, Presley, CA
, Howell, CR
, Agne, AA
, and Cherrington, AL
. The mediating role of self-efficacy in the association between diabetes education and support and self-care management. Health Educ Behav. (2022) 49:689–96. doi: 10.1177/10901981211008819
PubMed Abstract | Crossref Full Text | Google Scholar
link







